Nuclear Chemistry
An Introduction
Traditional chemical reactions occur as a result of the interaction betweenvalence electrons around an atom's nucleus (see our Chemical Reactionsmodule for more information). In 1896, Henri Becquerel expanded the field of chemistry to include nuclear changes when he discovered that uranium emitted radiation. Soon after Becquerel's discovery, Marie Sklodowska Curie began studying radioactivity and completed much of the pioneering work on nuclear changes. Curie found that radiation was proportional to the amount of radioactive element present, and she proposed that radiation was a property of atoms (as opposed to a chemical property of a compound).Marie Curie was the first woman to win a Nobel Prize and the first person to win two (the first, shared with her husband Pierre and Becquerel for discovering radioactivity; the second for discovering the radioactive elements radium and polonium).
Radiation and nuclear reactions
In 1902, Frederick Soddy proposed the theory that "radioactivity is the result of a natural change of an isotope of one element into an isotope of a different element." Nuclear reactions involve changes in particles in an atom's nucleus and thus cause a change in the atom itself. All elements heavier than bismuth (Bi) (and some lighter) exhibit natural radioactivity and thus can "decay" into lighter elements. Unlike normal chemical reactions that form molecules, nuclear reactions result in the transmutation of one element into a different isotope or a different element altogether (remember that the number of protons in an atom defines the element, so a change in protons results in a change in the atom). There are three common types of radiation and nuclear changes:
- Alpha Radiation (α) is the emission of an alpha particle from an atom'snucleus. An α particle contains two protons and two neutrons (and is similar to a He nucleus:
 ). When an atom emits an a particle, the atom's atomic mass will decrease by four units (because two protons and two neutrons are lost) and the atomic number (z) will decrease by two units. The element is said to "transmute" into another element that is two z units smaller. An example of an a transmutation takes place when uranium decays into the element thorium (Th) by emitting an alpha particle, as depicted in the following equation:
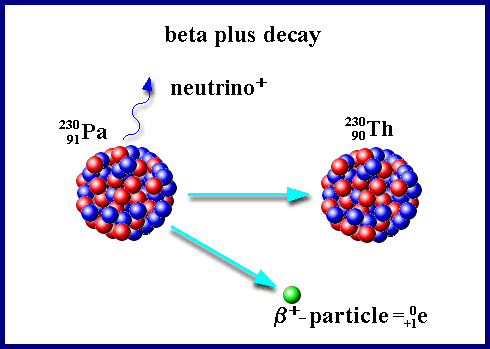
). When an atom emits an a particle, the atom's atomic mass will decrease by four units (because two protons and two neutrons are lost) and the atomic number (z) will decrease by two units. The element is said to "transmute" into another element that is two z units smaller. An example of an a transmutation takes place when uranium decays into the element thorium (Th) by emitting an alpha particle, as depicted in the following equation: - Beta Radiation (β) is the transmutation of a neutron into a proton and an electron (followed by the emission of the electron from the atom's nucleus:
 ). When an atom emits a β particle, the atom's mass will not change (since there is no change in the total number of nuclear particles), however the atomic number will increase by one (because the neutron transmutated into an additional proton). An example of this is the decay of the isotope of carbon named carbon-14 into the element nitrogen:
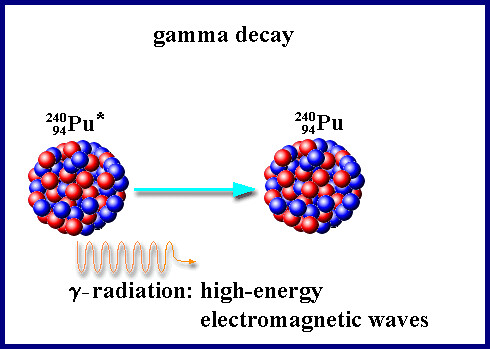
). When an atom emits a β particle, the atom's mass will not change (since there is no change in the total number of nuclear particles), however the atomic number will increase by one (because the neutron transmutated into an additional proton). An example of this is the decay of the isotope of carbon named carbon-14 into the element nitrogen: - Gamma Radiation (γ) involves the emission of electromagnetic energy(similar to light energy) from an atom's nucleus. No particles are emitted during gamma radiation, and thus gamma radiation does not itself cause the transmutation of atoms, however γ radiation is often emitted during, and simultaneous to, α or β radioactive decay. X-rays, emitted during the beta decay of cobalt-60, are a common example of gamma radiation.
92 | 2 | + | 90 |
6 | 0 -1 | + | 7 |
Half-life
Radioactive decay proceeds according to a principal called the half-life. The half-life (T½) is the amount of time necessary for one-half of the radioactive material to decay. For example, the radioactive element bismuth (210Bi) can undergo alpha decay to form the element thallium (206Tl) with a reaction half-life equal to five days. If we begin an experiment starting with 100 g of bismuth in a sealed lead container, after five days we will have 50 g of bismuth and 50 g of thallium in the jar. After another five days (ten from the starting point), one-half of the remaining bismuth will decay and we will be left with 25 g of bismuth and 75 g of thallium in the jar. As illustrated, the reaction proceeds in halfs, with half of whatever is left of the radioactive element decaying every half-life period.
| |
Radioactive Decay of Bismuth-210 (T½ = 5 days) |
The fraction of parent material that remains after radioactive decay can be calculated using the equation:
| Fraction remaining = | 1 2n | (where n = # half-lives elapsed) |
The amount of a radioactive material that remains after a given number of half-lives is therefore:
| Amount remaining = Original amount * Fraction remaining |
The decay reaction and T½ of a substance are specific to the isotope of the element undergoing radioactive decay. For example, Bi210 can undergoa decay to Tl206 with a T½ of five days. Bi215, by comparison, undergoes bdecay to Po215 with a T½ of 7.6 minutes, and Bi208 undergoes yet another mode of radioactive decay (called electron capture) with a T½ of 368,000 years!
Stimulated nuclear reactions
While many elements undergo radioactive decay naturally, nuclear reactions can also be stimulated artificially. Although these reactions also occur naturally, we are most familiar with them as stimulated reactions. There are two such types of nuclear reactions:
1. Nuclear fission: reactions in which an atom's nucleus splits into smaller parts, releasing a large amount of energy in the process. Most commonly this is done by "firing" a neutron at the nucleus of an atom. The energy of the neutron "bullet" causes the target element to split into two (or more) elements that are lighter than the parent atom.
| |
The Fission Reaction of Uranium-235 |
The Fission of U235
Concept simulation - Illustrates a nuclear fission reaction.
(Flash required)
During the fission of U235, three neutrons are released in addition to the two daughter atoms. If these released neutrons collide with nearby U235 nuclei, they can stimulate the fission of these atoms and start a self-sustaining nuclear chain reaction. This chain reaction is the basis of nuclear power. As uranium atoms continue to split, a significant amount of energy is released from the reaction. The heat released during this reaction is harvested and used to generate electrical energy.
Two Types of Nuclear Chain Reactions
Concept simulation - Reenacts controlled and uncontrolled nuclear chain reactions.
2. Nuclear fusion: reactions in which two or more elements "fuse" together to form one larger element, releasing energy in the process. A good example is the fusion of two "heavy" isotopes of hydrogen (deuterium: H2 and tritium: H3) into the element helium.
| |
Nuclear Fusion of Two Hydrogen Isotopes |
Nuclear Fusion
Concept simulation - Reenacts the fusion of deuterium and tritium inside of a tokamak reactor.
Fusion reactions release tremendous amounts of energy and are commonly referred to as thermonuclear reactions. Although many people think of the sun as a large fireball, the sun (and all stars) are actually enormous fusion reactors. Stars are primarily gigantic balls of hydrogen gas under tremendous pressure due to gravitational forces. Hydrogen molecules are fused into helium and heavier elements inside of stars, releasing energy that we receive as light and heat.
Atomic Theory II
Ions, Isotopes and Electron Shells
by Anthony Carpi, Ph.D.
In Atomic Theory I: The Early Days (see our Atomic Theory I module), we learned about the basic structure of the atom. Normally, atoms contain equal numbers of protons and electrons. Because the positive and negative charges cancel each other out, atoms are normally electrically neutral. But, while the number of protons is always constant in any atom of a givenelement, the number of electrons can vary.
Ions
When the number of electrons changes in an atom, the electrical chargechanges. If an atom gains electrons, it picks up an imbalance of negatively charged particles and therefore becomes negative. If an atom loses electrons, the balance between positive and negative charges is shifted in the opposite direction and the atom becomes positive. In either case, the magnitude (+1, +2, -1, -2, etc.) of the electrical charge will correspond to the number of electrons gained or lost. Atoms that carry electrical charges are called ions (regardless of whether they are positive or negative). For example, the animation below shows a positive hydrogen ion (which has lost an electron) and a negative hydrogen ion (which has gained an extra electron). The electrical charge on the ion is always written as a superscript after the atom's symbol, as seen in the animation.
Hydrogen Ion Simulation
Isotopes
The number of neutrons in an atom can also vary. Two atoms of the sameelement that contain different numbers of neutrons are called isotopes. For example, normally hydrogen contains no neutrons. An isotope of hydrogen does exist that contains one neutron (commonly called deuterium). Theatomic number (z) is the same in both isotopes; however the atomic massincreases by one in deuterium as the atom is made heavier by the extra neutron.
Hydrogen Isotope Simulation
Electron Shells
Ernest Rutherford's view of the atom consisted of a dense nucleussurrounded by freely spinning electrons (see our Atomic Theory I module). In 1913, the Danish physicist Niels Bohr proposed yet another modification to the theory of atomic structure based on a curious phenomenon called line spectra.
When matter is heated, it gives off light. For example, turning on an ordinary light bulb causes an electric current to flow through a metal filament that heats the filament and produces light. The electrical energyabsorbed by the filament excites the atoms' electrons, causing them to "wiggle". This absorbed energy is eventually released from the atoms in the form of light.
When normal white light, such as that from the sun, is passed through a prism, the light separates into a continuous spectrum of colors:
| |
Figure 1: Continuous (white light) spectra |
Bohr knew that when pure elements were excited by heat or electricity, they gave off distinct colors rather than white light. This phenomenon is most commonly seen in modern-day neon lights, tubes filled with gaseous elements (most commonly neon). When an electric current is passed through the gas, a distinct color (most commonly red) is given off by the element. When light from an excited element is passed through a prism, only specific lines (or wavelengths) of light can be seen. These lines of light are called line spectra. For example, when hydrogen is heated and the light is passed through a prism, the following line spectra can be seen:
| |
Figure 2: Hydrogen line spectra |
Each element has its own distinct line spectra. For example:
| |
Figure 3: Helium line spectra |
| |
Figure 4: Neon line spectra |
To Bohr, the line spectra phenomenon showed that atoms could not emitenergy continuously, but only in very precise quantities (he described the energy emitted as quantized). Because the emitted light was due to the movement of electrons, Bohr suggested that electrons could not move continuously in the atom (as Rutherford had suggested) but only in precise steps. Bohr hypothesized that electrons occupy specific energy levels. When an atom is excited, such as during heating, electrons can jump to higher levels. When the electrons fall back to lower energy levels, precise quanta of energy are released as specific wavelengths (lines) of light.
Under Bohr's theory, an electron's energy levels (also called electron shells) can be imagined as concentric circles around the nucleus. Normally, electrons exist in the ground state, meaning they occupy the lowest energy level possible (the electron shell closest to the nucleus). When an electron is excited by adding energy to an atom (for example, when it is heated), the electron will absorb energy, "jump" to a higher energy level, and spin in the higher energy level. After a short time, this electron will spontaneously "fall" back to a lower energy level, giving off a quantum of light energy. Key to Bohr's theory was the fact that the electron could only "jump" and "fall" to precise energy levels, thus emitting a limited spectrum of light. The animation linked below simulates this process in a hydrogen atom.
Bohr's Atom: Quantum Behavior in Hydrogen
Concept simulation - Reenacts electron's "jump" and "fall" to precise energy levels in a hydrogen atom.
occupy specific energylevels, he also predicted that those levels had limits to the number of electrons each could hold. Under Bohr's theory, the maximum capacity of the first (or innermost) electron shell is two electrons. For any element with more than two electrons, the extra electrons will reside in additional electron shells. For example, in the ground state configuration of lithium (which has three electrons) two electrons occupy the first shell and one electron occupies the second shell. This is illustrated in the animation linked below.
The Lithium atom
For further details, the table linked below shows the electron configurations of the first eleven elements.