Chapter 1: The atom
( Study Physics Online )
An atom consists of a positive charged atomic nucleus where you can find protons and neutrons and it consists of a negative charged atomic shell with electrons. In every atom the number of the electrons is equal to the number of the protons so it is neutral. The number of the protons decides which chemical element the atom is. The first element in the "Periodic table of the elements" is hydrogen. The elements in the "Periodic table of the elements" are sorted by the number of the protons. The atomic nucleus of a hydrogen atom consists of only one proton. But there are a few isotops of every element. Isotops are atoms with the same number of protons, but another number of neutrons. The different isotops of one element do not differ in their chemical properties. There are for example three isotops of hydrogen. The first isotop is the one I wrote about. The second isotop of hydrogen is deutrium with one proton and one neutron in his atomic nucleus and the third isotop is tritium which has got one proton and two neutrons in his atomic nucleus. In the atomic nucleus of a tritiumatom there is no balance between the protons and the neutrons so it is instable and decays. The particle which is emited from this decay is radioaktiv and it is charged. You can make ions of atoms. We can say that an ion is an atom which has got less or more electrons than protons. An ion is not neutral an so it is radioactif.
Chapter 2: Radioactivity
Radioactivity means that atoms decays. The reason for this decays is that they are instable. A atomic nucleus is instable when he is to heavy or when a balance is missing between the protons and the neutrons. Every atom which has got a higher number of nucleons (protons and neutrons togehter) than 210 is instable. There are three types of decays: alpha decay, beta decay and gamma decay. Because it is impossible today to say which atomic nucleus will be the next who decays there statistics. We can say how many atomic nucleus will decay in a certain time. This is the princip for half lifes. After one half life a half of the atomic nucleus of a certain material decayed. Plutonium-239 for example has got a half life 24,000 years, radium-228 has got a half life of 6.7 years, thorium-232 has got a half life of 14,000,000,000 years and polonium-212 has got a half life 0.0000003 seconds. There are many physical properties, but I will talk about the acivity now. The activity is the number of decays devided by a certain time. the unit of the activity is becquerel. 1 becquerel is one decay per second. So 20 becquerels are 20 decays per second. To prove these decays there is a geiger counter. It consists of a closed tube which is often filled with argon. At the end of the tube there is a wire, which is not allowed to touch the other end of the tube or the walls. The wire is charged positive and the walls are charged negative. A radioactive particle which flows into the tube ionizes one or a few gas atoms. The out-pushed electrons go to the wire. The consequence is a voltage surge. This voltage surge is shown on an output device as a decay. On the photo there shown a geiger counter.
The alpha decay
When we talk about the alpha decay then it means that a twice positive charged heliumion (helium atomic nucleus) is emited from the atomic nucleus. Then we find two protons ans two neutrons less in this atomic nucleus, so it is lighter. The alpha radiation is the most dangerous of the three types of radiation, but a sheet of paper is enough to protect oneself. The skin protects us also from alpha radiation.
The beta minus decay
There are two types of the beta decay. The one is the beta minus decay and the other is the beta plus decay. When we talk about the beta minus decay a neutron decays into a proton, an electron and an antineutrino. The electron and the antineutrino are emited. The radioactive particle is the electron. The number of nucleons do not change, but we have got one proton more than before the decay. 2 or 3 cm of wood are enough to protect oneself.
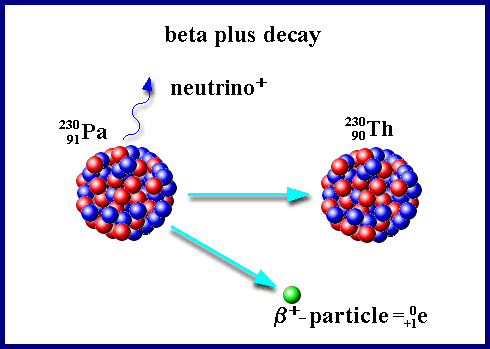
The beta plus decay
When we talk about the beta plus decay a proton decays into a neutron, a positron (the antiparticle of the electron) and a neutrino. The positron and the neutrino are emited. The radioactive particle is the positron.
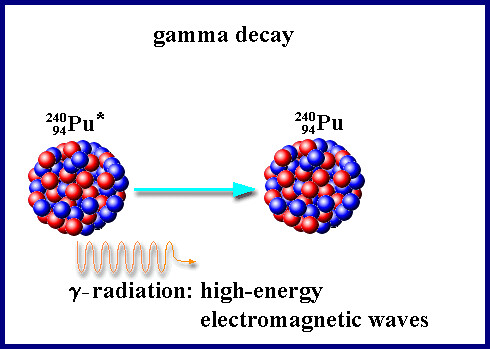
The gamma decay
When we talk about the gamma decay high-energy electromagnetic waves are emited from the atomic nucleus. This waves are photons, which have got a higher frequency and less wave long than light. A gamma decay can happen after an alpha decay or a beta decay, because the atomic nucleus is very energitif. You need a big wall of lead to protect yourself from gamma radiation.
Chapter 3: The applications of radioactivity
Everyone knows that strong radiation is not good fot the health, but we use radioactive materials for nuclear power plants ans nuclear weapons (Chapter 4) for example. But there are good sides for radioactivity, too. There for example nuclear medicine. An X-ray instrument sends X-Rays throught our body onto a photo plate. Where the photo plate becomes black the X-rays goes throught our body, there where the photo plate stays transparent the X-rays do not pass our body. Another positive aspect is the radiotherapy. It is used to destroy cancer. In old clocks which have illuminated you can find radium and thorium which were used to bring the zinc sulfite to illuminate. The glowing trunk for camping lamps contained thorium. The energy source for the batteries for cardiac pacemaker is plutonium-238. There is not any nuclear fission in those batteries, because the energy source is the natural nuclear decay. Radionuclide batteries are also used for space probes like Voyager I, Voyager II and Cassini who are very long in space and so they need radionuclide batteries who are an energy source for a long time. In the next chapter I will talk bout nuclear power plants and nuclear weapons.
Chapter 4: Nuclear reactions and their applications
There are many nuclear reactions, but I will only discribe the nuclear fission and the nuclear fusion. For a nuclear fission in a nuclear power plant or for an explosion of a nuclear bomb you need plutonium-239 or uranium-235 as a split material. To make a nuclear fission it is necessary to bombard the split material with thermal neutrons. After the fission there there are two new atoms and and two or three free neutrons. This free neutrons make a fission of other atoms and so it is a nuclear chain reaction.
The animation of a nuclear fission:
In a nuclear bomb there is a globe made of plutonium-239 or uranium-235. In this globe there is a neutron source which only effective when the TNT (trinitrotoluene) exploses. Because of the compression of the explosion the critical mass of the split material is overstepped. There are nuclear bomb which are build otherwise, but the princip is always the same. This both materials are very expensive, because on earth we find very little plutonium so it means that we must produce plutonium. To produce plutonium it is necessary to bombard the natural and very cheap uranium-238 with neutrons to make uranium-239. Uranium-239 decays to neptunium-239 and neptunium-239 decays after a certain time to plutonium-239. You can find uranium-235 in nature, but only in uranium-238. To split this uranium-235 from uranium-238 is very expensive, because their chemical properties are the same so it is not possible to split them in a chemical way. A nuclear bomb like this can have an explosion force of 20 kilotons (20000 tons). This means that an explosion of such a bomb is as effective as the explosion of 20 kilotons of TNT.
Hydrogen bombs can reach an explosion force of 20 megatons (20 million tons). This bombs are also knows as three-phase fuzes. The fission like in a nuclear bomb is only the first phase. In the second phase there is a fusion between deutrium and tritium. The temperatur in the second phase behave 200 to 300 million degrees celsius (much hoter than the core of the sun). The third phase is the fission of uranium-238 which is of the outer side of the bomb. Under this conditions the fission of uranium-238 is possible. The princip of power plants is the same like in nuclear bombs, but without using TNT. The reason why nuclear power plants do not exploses is that there are control rods to control the number of the neutrons in the reactor. This is a controlled nuclear chain reaction in the opposite of an uncontrolled nuclear chain reaction in nuclear bombs. The nuclear power plants in the future will be fusion reactors which do not crack heavy atomic nucleus, but fuses light atomic nucleus. Fusion are today possible but energy which you need for a fusion is higher than the energy you get and this is not the sense of nuclear fusions. With fusions the last elements of the "Periodic table of the elements" have been created, because their are not on earth. In 1999 a few physicists thought that they have discovered the element 118 but two years later in 2001 they said that it was a mistake, so element 114 is the last know element. In stars there are also fusions. In our sun it is the proton proton cycle which you can find on the website of astronomy and astrophysics. Now I will give an answer why we get energy from this nuclear reactions. We must begin which Einstein's famous formula: E=mc2 (E stands for energy, m stands for mass and c stands for the speed of light in the vacuum). This formula makes it possible transform masse in energy. Atomic nucleus have got different binding energy. The binding energy is the energy which holds the nucleons together. Because of this fact there is in every atomic nucleus a mass defect. A free proton and a free neutrons weighs more than deutrium (heavy hydrgen, consists of one proton and one neutron). Iron has got the highest binding energy and stands in the middle of the "Periodic table of the elements". When somebody goes closer to this middle with fissions or fusions a part will be transformed into energy.
The animation of a nuclear fusion:
( Study Physics Online )
An atom consists of a positive charged atomic nucleus where you can find protons and neutrons and it consists of a negative charged atomic shell with electrons. In every atom the number of the electrons is equal to the number of the protons so it is neutral. The number of the protons decides which chemical element the atom is. The first element in the "Periodic table of the elements" is hydrogen. The elements in the "Periodic table of the elements" are sorted by the number of the protons. The atomic nucleus of a hydrogen atom consists of only one proton. But there are a few isotops of every element. Isotops are atoms with the same number of protons, but another number of neutrons. The different isotops of one element do not differ in their chemical properties. There are for example three isotops of hydrogen. The first isotop is the one I wrote about. The second isotop of hydrogen is deutrium with one proton and one neutron in his atomic nucleus and the third isotop is tritium which has got one proton and two neutrons in his atomic nucleus. In the atomic nucleus of a tritiumatom there is no balance between the protons and the neutrons so it is instable and decays. The particle which is emited from this decay is radioaktiv and it is charged. You can make ions of atoms. We can say that an ion is an atom which has got less or more electrons than protons. An ion is not neutral an so it is radioactif.
Chapter 2: Radioactivity
Radioactivity means that atoms decays. The reason for this decays is that they are instable. A atomic nucleus is instable when he is to heavy or when a balance is missing between the protons and the neutrons. Every atom which has got a higher number of nucleons (protons and neutrons togehter) than 210 is instable. There are three types of decays: alpha decay, beta decay and gamma decay. Because it is impossible today to say which atomic nucleus will be the next who decays there statistics. We can say how many atomic nucleus will decay in a certain time. This is the princip for half lifes. After one half life a half of the atomic nucleus of a certain material decayed. Plutonium-239 for example has got a half life 24,000 years, radium-228 has got a half life of 6.7 years, thorium-232 has got a half life of 14,000,000,000 years and polonium-212 has got a half life 0.0000003 seconds. There are many physical properties, but I will talk about the acivity now. The activity is the number of decays devided by a certain time. the unit of the activity is becquerel. 1 becquerel is one decay per second. So 20 becquerels are 20 decays per second. To prove these decays there is a geiger counter. It consists of a closed tube which is often filled with argon. At the end of the tube there is a wire, which is not allowed to touch the other end of the tube or the walls. The wire is charged positive and the walls are charged negative. A radioactive particle which flows into the tube ionizes one or a few gas atoms. The out-pushed electrons go to the wire. The consequence is a voltage surge. This voltage surge is shown on an output device as a decay. On the photo there shown a geiger counter.
The alpha decay
When we talk about the alpha decay then it means that a twice positive charged heliumion (helium atomic nucleus) is emited from the atomic nucleus. Then we find two protons ans two neutrons less in this atomic nucleus, so it is lighter. The alpha radiation is the most dangerous of the three types of radiation, but a sheet of paper is enough to protect oneself. The skin protects us also from alpha radiation.
The beta minus decay
There are two types of the beta decay. The one is the beta minus decay and the other is the beta plus decay. When we talk about the beta minus decay a neutron decays into a proton, an electron and an antineutrino. The electron and the antineutrino are emited. The radioactive particle is the electron. The number of nucleons do not change, but we have got one proton more than before the decay. 2 or 3 cm of wood are enough to protect oneself.
The beta plus decay
When we talk about the beta plus decay a proton decays into a neutron, a positron (the antiparticle of the electron) and a neutrino. The positron and the neutrino are emited. The radioactive particle is the positron.
When we talk about the gamma decay high-energy electromagnetic waves are emited from the atomic nucleus. This waves are photons, which have got a higher frequency and less wave long than light. A gamma decay can happen after an alpha decay or a beta decay, because the atomic nucleus is very energitif. You need a big wall of lead to protect yourself from gamma radiation.
Chapter 3: The applications of radioactivity
Everyone knows that strong radiation is not good fot the health, but we use radioactive materials for nuclear power plants ans nuclear weapons (Chapter 4) for example. But there are good sides for radioactivity, too. There for example nuclear medicine. An X-ray instrument sends X-Rays throught our body onto a photo plate. Where the photo plate becomes black the X-rays goes throught our body, there where the photo plate stays transparent the X-rays do not pass our body. Another positive aspect is the radiotherapy. It is used to destroy cancer. In old clocks which have illuminated you can find radium and thorium which were used to bring the zinc sulfite to illuminate. The glowing trunk for camping lamps contained thorium. The energy source for the batteries for cardiac pacemaker is plutonium-238. There is not any nuclear fission in those batteries, because the energy source is the natural nuclear decay. Radionuclide batteries are also used for space probes like Voyager I, Voyager II and Cassini who are very long in space and so they need radionuclide batteries who are an energy source for a long time. In the next chapter I will talk bout nuclear power plants and nuclear weapons.
Chapter 4: Nuclear reactions and their applications
There are many nuclear reactions, but I will only discribe the nuclear fission and the nuclear fusion. For a nuclear fission in a nuclear power plant or for an explosion of a nuclear bomb you need plutonium-239 or uranium-235 as a split material. To make a nuclear fission it is necessary to bombard the split material with thermal neutrons. After the fission there there are two new atoms and and two or three free neutrons. This free neutrons make a fission of other atoms and so it is a nuclear chain reaction.
The animation of a nuclear fission:
In a nuclear bomb there is a globe made of plutonium-239 or uranium-235. In this globe there is a neutron source which only effective when the TNT (trinitrotoluene) exploses. Because of the compression of the explosion the critical mass of the split material is overstepped. There are nuclear bomb which are build otherwise, but the princip is always the same. This both materials are very expensive, because on earth we find very little plutonium so it means that we must produce plutonium. To produce plutonium it is necessary to bombard the natural and very cheap uranium-238 with neutrons to make uranium-239. Uranium-239 decays to neptunium-239 and neptunium-239 decays after a certain time to plutonium-239. You can find uranium-235 in nature, but only in uranium-238. To split this uranium-235 from uranium-238 is very expensive, because their chemical properties are the same so it is not possible to split them in a chemical way. A nuclear bomb like this can have an explosion force of 20 kilotons (20000 tons). This means that an explosion of such a bomb is as effective as the explosion of 20 kilotons of TNT.
Hydrogen bombs can reach an explosion force of 20 megatons (20 million tons). This bombs are also knows as three-phase fuzes. The fission like in a nuclear bomb is only the first phase. In the second phase there is a fusion between deutrium and tritium. The temperatur in the second phase behave 200 to 300 million degrees celsius (much hoter than the core of the sun). The third phase is the fission of uranium-238 which is of the outer side of the bomb. Under this conditions the fission of uranium-238 is possible. The princip of power plants is the same like in nuclear bombs, but without using TNT. The reason why nuclear power plants do not exploses is that there are control rods to control the number of the neutrons in the reactor. This is a controlled nuclear chain reaction in the opposite of an uncontrolled nuclear chain reaction in nuclear bombs. The nuclear power plants in the future will be fusion reactors which do not crack heavy atomic nucleus, but fuses light atomic nucleus. Fusion are today possible but energy which you need for a fusion is higher than the energy you get and this is not the sense of nuclear fusions. With fusions the last elements of the "Periodic table of the elements" have been created, because their are not on earth. In 1999 a few physicists thought that they have discovered the element 118 but two years later in 2001 they said that it was a mistake, so element 114 is the last know element. In stars there are also fusions. In our sun it is the proton proton cycle which you can find on the website of astronomy and astrophysics. Now I will give an answer why we get energy from this nuclear reactions. We must begin which Einstein's famous formula: E=mc2 (E stands for energy, m stands for mass and c stands for the speed of light in the vacuum). This formula makes it possible transform masse in energy. Atomic nucleus have got different binding energy. The binding energy is the energy which holds the nucleons together. Because of this fact there is in every atomic nucleus a mass defect. A free proton and a free neutrons weighs more than deutrium (heavy hydrgen, consists of one proton and one neutron). Iron has got the highest binding energy and stands in the middle of the "Periodic table of the elements". When somebody goes closer to this middle with fissions or fusions a part will be transformed into energy.
The animation of a nuclear fusion:








Nice job done here, As I found nuclear physics little difficult but through the post I am able to get some confidence back in these subjects
ReplyDeleteOnline Math Tutors.
Cheers for the collection. Very handy. Thanks again.
ReplyDeleteSam
http://www.zeuscreative.co.uk
I appreciate that you have shared this informative reviews. I'm so pleased to have cross over on this informative post. Fleece jacket
ReplyDeleteThis post is a monster of information. Learning nuclear physics on a website! I would rather go back to college.
ReplyDeleteNice post!!very informative...thanks for sharing..
ReplyDeleteNGOs working for women empowerment in Noida
Hmm - I was looking for a good article explaining atoms for my elementary school age kids. This would be a bit beyond them. Do you have other suggestions? Thanks!
ReplyDeleteMy Homeschool Blog
Great blog! I enjoy learning from your informative posts, thanks and keep on updating your useful posts!
ReplyDeleteלימודי מנהל עסקים תואר ראשון
Great blog and post, keep it up i will be subscribing to your feed!
ReplyDeleteHigh School Diploma Online
I would like to say that this post is awesome, nice written and include almost all important information. I'd like to see more posts like this
ReplyDeleteWow, atomic structures and isotopes always sounded complicated, but this breakdown makes it much clearer. Pay Someone To Take My Online Course can be a great solution if you're struggling with chemistry concepts like ions and radioactive decay. I remember having a tough time with these topics in school. Sometimes, getting extra help for science subjects is a game-changer. If you’re taking an online chemistry course and feeling stuck, I totally get it! There’s no harm in finding someone who can guide you through the tougher topics.
ReplyDeleteThis paragraph offers a great overview of radioactivity and detection methods like the Geiger counter. For students who find topics like these challenging, our online exam help service is here to support you. We provide expert guidance to help you understand difficult concepts and perform better in your online exams with confidence.
ReplyDelete